
Since the effective nuclear charge also increases from left to right, then the attraction between the protons and electrons are greater therefore a higher ionization energy is required to remove the electron. If the orbitals are fully filled or half filled then it will take more energy to remove an electron from them. By determining which period the element is located within. Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. This is the general trend but there are many exceptions based on how the orbitals are filled. Whichever portion of the table the element falls under will show its valence electron sub-shell. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. Both increase from left to right and decrease going from top to bottom. The increased numbers of electrons in the valence shell, also have an increased number of equivalent protons, and they will be pulled in tighter and therefore. Atomic radius, which is half the distance between the. Ionization energy and electronegativity have the same trend. These electrons at the higher valence shell are farther away from the nucleus, increasing atomic radius. The more protons there are means that they attract the electrons more, thus making the atomic radius smaller. Atomic radius decreases going across the periodic table because the effective nuclear charge increases. How do they relate to shielding effect and number of electrons or protons?Ītomic radius increases going down the periodic table because a valence electron is added into a new energy level and the core electrons repel each other (shielding effect) which contribute to the atomic radius increasing. Kristen Kim 1I wrote:Can someone explain the reasons behind the trends we see in the periodic table? decreases.Īcross a period, effective nuclear charge increases and valence electrons are more strongly attracted to the increasingly positive nucleus so it is harder to remove a valence electron from the atom. For main group elements, the number of valence electrons usually ranges between 1 and 8 because eight electrons forms a complete octet. The group number gives the number of valence electrons in an element. Down a group, valence electrons are further away from the nucleus and less tightly bound hence less energy is required to remove a valence electron from the atom and first I.E. Elements with similar physical and chemical properties belong in a group. First ionization energy generally increases across a period and decreases down a group. Going across a row on the periodic table, left to right, the trend is different. This is because the valence electron shell is getting larger and there is a larger principal quantum number, so the valence shell lies physically farther away from the nucleus. Shielding effect remains relatively the same across a period because electrons are added to the same shell while nuclear charge increases. As you go down a column of the periodic table, the atomic radii increase. So the atom becomes more compact and the atomic radius decreases. Down a group, valence electrons are in shells that are further away from the nucleus and hence atomic radius increases down the group.Īcross a period there is a decrease because effective nuclear charge increases and valence electrons are more strongly attracted to the nucleus. Atomic radius generally increases down a group and decreases across a period. Addition of another electron does not result in a fractional decrease in the electrostatic attraction to any given electron, but it does increase the electron-electron repulsion, so an overall decrease in Net attractive force.1. The periodic trend of the number of valence electrons being linked to an elements group number only works for Groups 1-2 and 13-18. The internal energy levels “shield” and reduce electrostatic attraction of the valence electrons to the protons. (2) Number of energy levels: The greater the number of energy levels, the larger the atomic radii. The new energy shells provide shielding, allowing the valence.  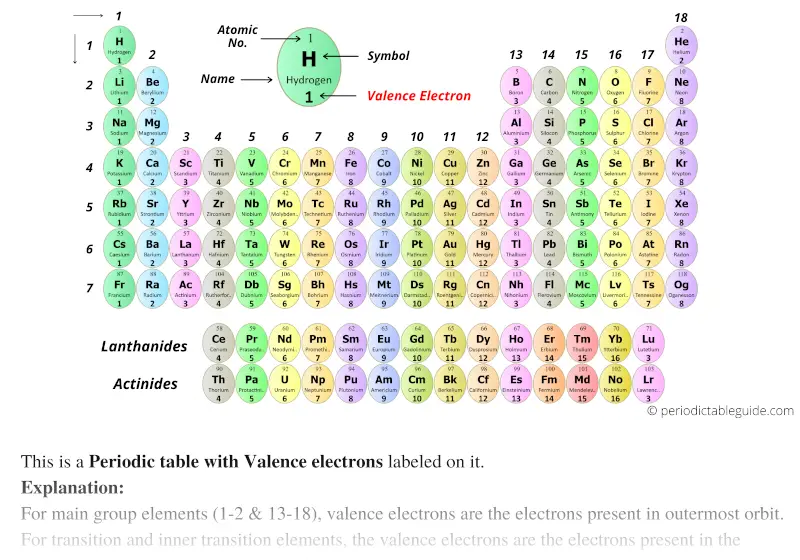
(1) Nuclear charge (number of protons) : The stronger the ‘pull’ the protons have to the electrons with electrostatic attraction, then the smaller the size of the atom radii Once again protons are added moving down a group, but so are new energy shells of electrons. Bohr models for the first three periods of the periodic table are shown below. Atomic Radii is affected by two main factors :
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
